Understanding the Definitions of Ionization Energy and Electron Affinity
H g H g e -. Consider the following data for chlorine.
Solved O Electronic Structure Understanding The Definitions Chegg Com
Definition of electron affinity Explanation of electron affinity.
. Its electronegativity is 1 28 2 30 5 3 3. The main difference between electron affinity and ionization energy is that electron affinity gives the amount of energy released when an atom gains an electron whereas ionization energy is the amount of energy required to remove an. Objectives Examine periodic trends in ionization energy Examine periodic trends in electron affinity Key Terms Ionization energy Electron Affinity Ionization Energy Ease at which electrons can be removed from an atom or ion First ionization energy I1 is the energy required to remove the first electron from neutral atom.
Ionization energy also called ionization potential is the energy necessary to remove an electron from the neutral atom. Ionization reactions always absorb energy called the ionization energy IE. Electron affinity the energy required to add 1 mole of electrons to 1 mole of gaseous atoms or ions.
A reaction in which a neutral atom loses an outer electron e- on the right side of the equation is called an ionization reaction. Compare atoms for their ability to form cations andor anions. Consider the following data for silver.
Ionization Energy Electron Affinity. Ionization energy is the amount of energy needed to remove an electron from a neutral atom. Consider the following data for potassium.
In other words it can be expressed as the neutral atoms likelihood of gaining an electron. Mol ionization energy 9470 kJ mol heat of fusion KJ 277 mol You may find additional useful data in. Does the following reaction absorb or release energy.
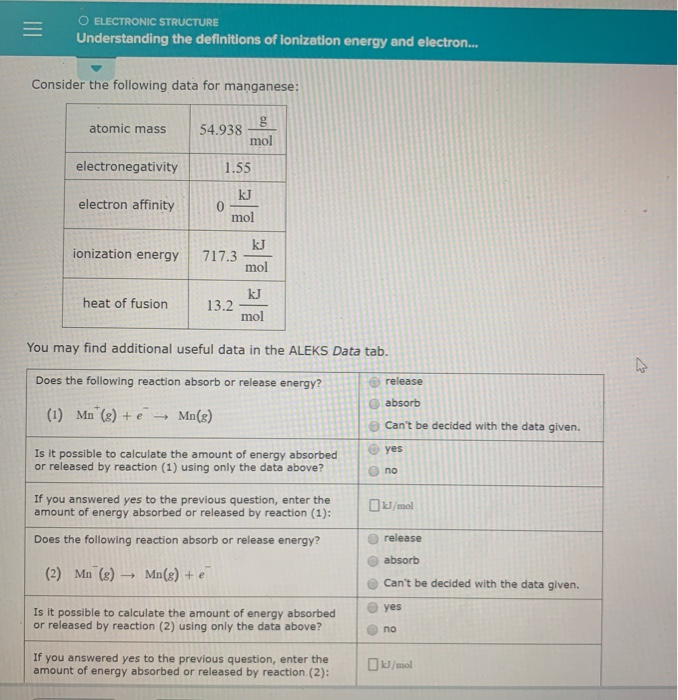
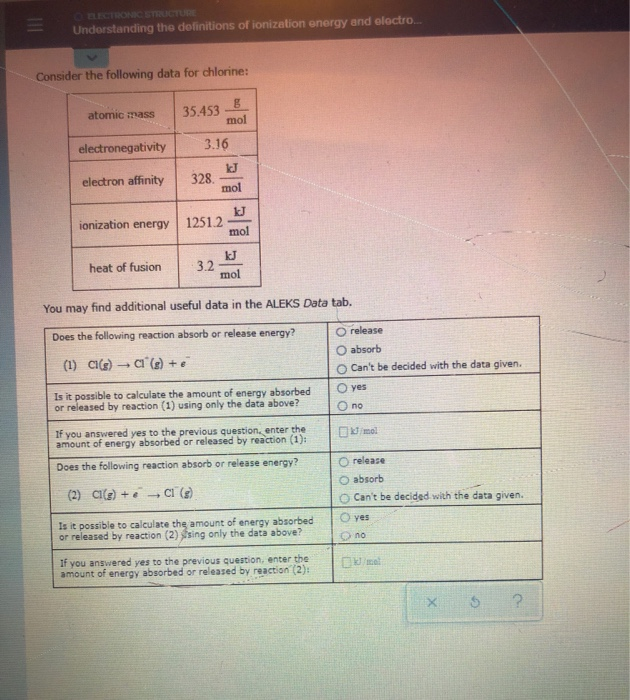
X energy X e where X is any atom or molecule capable of being ionized X is that atom or molecule with an electron removed positive ion and e is the removed electron. Atomic mass 354538 electronegativity 316 electron affinity 328. Atomic mass 74922 mol electronegativity 218 electron affinity kJ 78.
Understanding the definitions of ionization energy and electro. The more negative the electron affinity value the higher an atoms affinity for electronsThe energy of an atom is stated when an atom loses or gains energy through chemical reactions that cause the. Atomic mass g 39098 mol electronegativity 082 electron affinity kJ 484 mol ionization energy KJ 4188 mol kJ heat of fusion 233 mol You may find additional useful data in the ALEKS Data tab.
Click hereto get an answer to your question 56 174 The ionisation energy and electron affinity of an element are 130ev and 38ev respectively. Consider the following data for arsenic. Electronegativity the relative ability of an atom to attract an electron pair within a covalent bond.
O ELECTRONIC STRUCTURE Understanding the definitions of ionization energy and electron. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators. Atomic mass 10787 mol 193 electronegativity electron affinity 1256 mol ionization energy 7310 mol heat of fusion 113 mol You may find additional useful data in the ALEKS Data tab.
Understanding the definitions of ionization energy and electron. In chemistry and atomic physics the electron affinity of an atom or molecule is defined as. 4 40.
Atomic mass 26982 B mol electronegativity 161 kJ 425 mol electron affinity ionization energy 5775 mol heat of fusion 107 mol You may find additional useful data in the ALEKS Data tab. By definition the first ionization energy of an element is the energy needed to remove the outermost or highest energy electron from a neutral atom in the gas phase. The process by which the first ionization energy of hydrogen is measured would be represented by the following equation.
X e X energy Affinity H. E O ELECTRONC STRUCTURE Understanding the definitions of ionization energy and electron Consider the following data for aluminum. Electron affinities are more difficult to measure than ionization energies.
Electron affinity is a quantitative measurement of the tendency of an atom of an element to accommodate the electron in its outermost orbital. Chemistry Tutoring is a subscription service that provides unlimited access to engaging interactive chemistry tutoring modules practice worksheets and a proprietary library of chemistry analogies. Can anyone explain these to me in normal English please.
Ionization energy is related with making cations from neutral atoms and electron affinity is related with. Understanding the definitions of ionization energy and electron. Electron affinity is the amount of energy released when electron is added to an atom.
To get regular Just Like. I am having a hard time understanding these concepts from the definitions given in the book and from my teacher. Electro Negativity Ionization Energy and Electron Affinity.
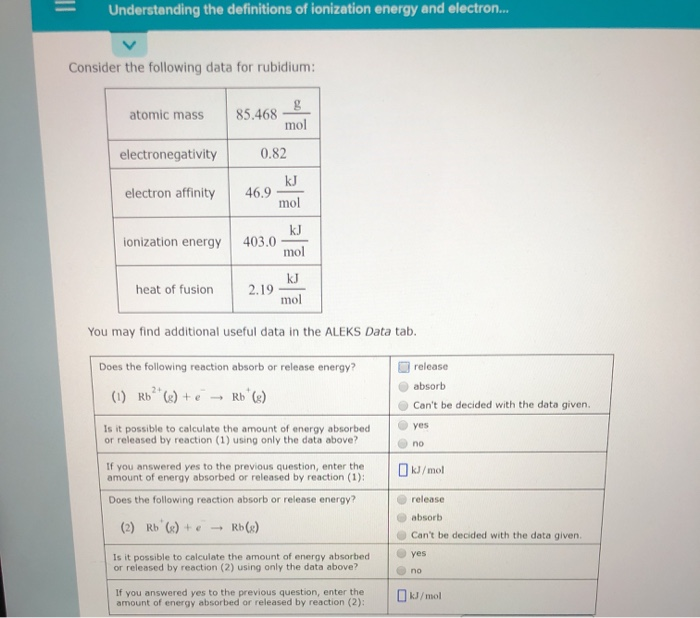
The change in energy in kJmole of a neutral atom or molecule in the gaseous phase when an electron is added to the atom to form a negative ion. Ionization Energy and Electron Affinity. Release Does the following reaction absorb or.
Electron affinity and ionization energy are two chemical terms used to describe the behavior of electrons and atom quantitatively. What is the difference between Ionization Energy and Electron Affinity. Electron affinity Electronegativity Ionization energy 1.
If the atom has a tendency to accept the electron then it will release the energy and electron affinity is represented by a negative sign. The energy released when an electron is added to a gaseous atom which is in its ground state to form a gaseous negative ion is defined as the first electron affinity. Electron affinity is defined as the quantitative measurement of the energy change that results from adding a new electron to a neutral atom or molecule in the gaseous state.
Note that ionization energies measure the tendency of a neutral atom to resist the loss of electrons. Ionisation energy the energy required to remove 1 mole of electrons from 1 mole of gaseous atoms or ions. Mol ionization energy 12512 mol heat of fusion 32 You may find additional useful data in the ALEKS Data tab.
Understanding the definitions of energy and electron affinity Problem.

Solved Understanding The Definitions Of Ionization Energy Chegg Com

8 3c Understanding The Definitions Of Ionization Energy And Electron Affinity Youtube
Solved Understanding The Definitions Of Ionization Energy Chegg Com
No comments for "Understanding the Definitions of Ionization Energy and Electron Affinity"
Post a Comment